Which of the Following Describes a Strong Acid
Hence visual c represents a weak acid. Select all that apply The Ka is very small Dissociates completely into ions in water Is a strong electrolyte.
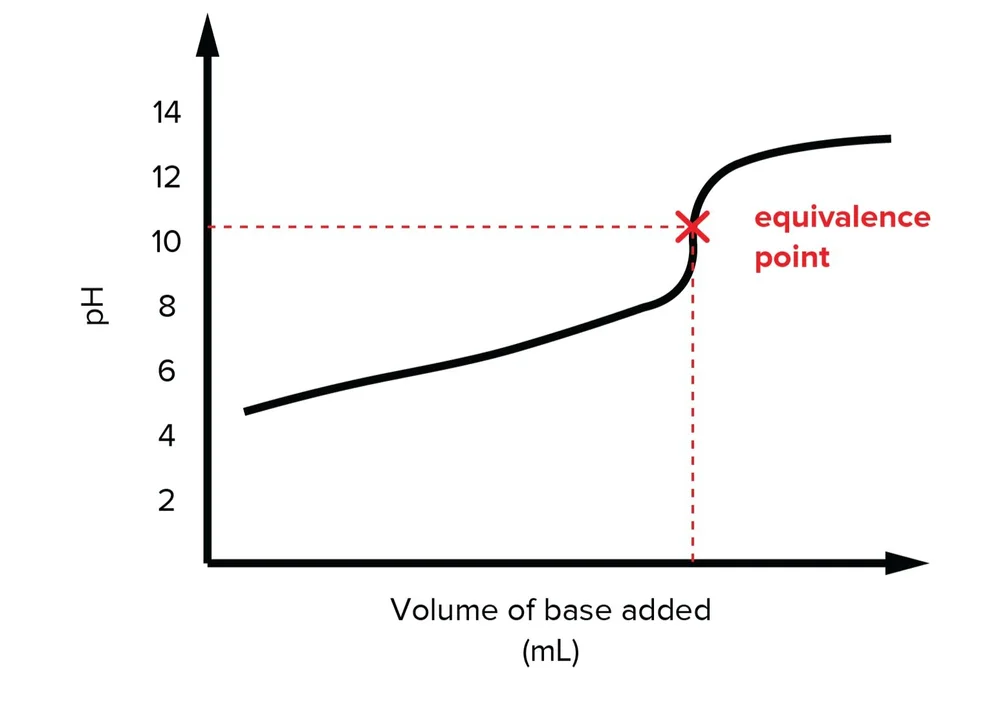
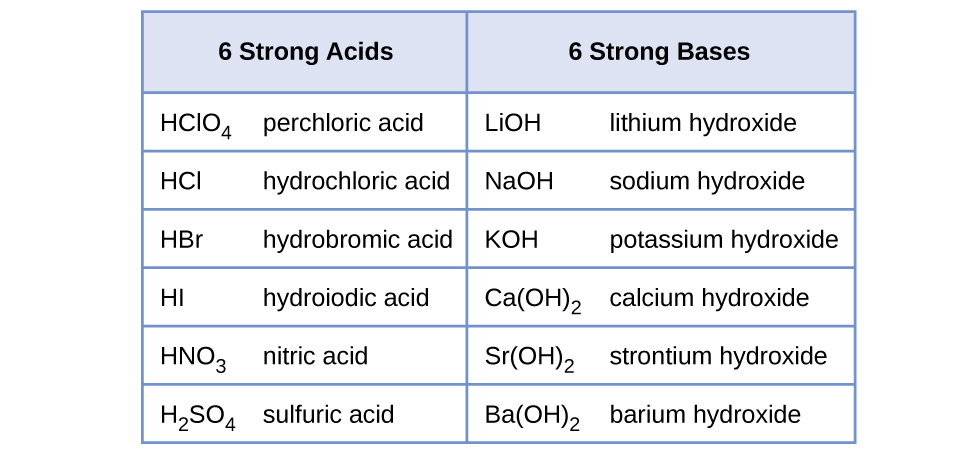
Acids And Bases For The Mcat Everything You Need To Know Shemmassian Academic Consulting
The degree to which it dissociates in water.

. 6 Which of the following statements describes what happens in a buffer solution when a strong acid is added. The pH of the solution is defined as the negative logarithm of the hydrogen ions concentration in an aqueous solution. A solution that has a pH of 2 is a strong acid solution.
Drain cleaner has a pH of about 14. Solved Which One Of The Following Is A Strong Acid Select Chegg Com. Presence versus absence of a halogen ion in the chemical structure C.
Complete versus partial ionization in solution. NaCl is the acid. It is a weak base.
A strong acid is any acid that ionizes completely in solutions. A substance in a solution that releases hydrogen ions and lowers the pH. The initial pH before the addition of any strong base is higher or less acidic than the titration of a strong acid.
Answerwith nitric acid or ethanoic acid. After each decay press the reset nucleus button to watch the process again. Strong acids and strong bases combine to form salts.
The H ions from the strong acid combine with basic ions in the buffer. In the titration of a strong acid with a strong base. How can it be classified based on its pH.
Calculate the pH of each of the following aqueous solutions. Chapter 16 Problem 29Q is solved. Negative versus positive pH D.
There are several characteristics that are seen in all titration curves of a weak acid with a strong base. Which of the following describes a phenol. E HClO2 H C l O 2.
If the pH of the solution is greater than 7. It is a very strong base. See answer 1 Best Answer.
Click card to see definition. Which one of the following best describes a strong acid. Which of the following curves corresponds to the titration of a weak base.
2018-02-05 12. Which of the following options correctly describe the behavior of this system when a strong acid is added to it. - 11501737 hazelmercader06 hazelmercader06 25022021 Science Junior High School answered Which of the following describes a phenol.
Tap card to see definition. A strong acid completely dissociates in solution A strong acid completely dissociates in solution At the same concentration which acid has the greater electrical conductivity nitric acid or ethanoic acid. Which of the following statements best describes why strong acids and strong bases are good electrolytes.
D H2CO3 H 2 C O 3. Strong acids and strong bases make water less pure. True False Which of the following pairs lists a substance that can neutralize CaOH2 and the salt that would be produced from the reaction.
Chemistry questions and answers. Enter your answers to two decimal places a 100 mL of 010 M NH 4 Cl plus 100 mL of 010 M NaOH 1098 1098 a solution containing a strong acid and a strong base. So the acid is partially ionized.
It is a strong base. BProduces hydronium ions in solution. Hence visual b represents strong acid such as.
Write a description of alpha decay for po-. An acid is a substance that donates H ions in a reaction. Nitric acid Nitric acid.
The H ions from the strong acid combine with acidic ions in the buffer. Ionizes completely in solution. NaOH is the acid.
Complete the following reaction and identify the Brønsted acidNaOHaq HClaq A. This means it gives off the greatest number of hydrogen ions or protons when placed in a solution. NCERT P Bahadur IIT.
It is a weak acid. Which of the following statements best describes what determines whether an acid is a strong acid or a weak acid. All of the Above.
Reacts with metals that are more active than hydrogen. If the pH of the solution is equal to 7 is said to be a neutral solution. Proton donation versus electron acceptance B.
Strong acids and strong bases neutralize each other. HCl is the acid. Is hydrochloric acid a strong acid.
In case of visual c there are three hydronium ions three anions of the corresponding acid and three unionized acid moles. NCERT DC Pandey Sunil Batra HC Verma Pradeep Errorless. Initial pH of acid no base has been addedHalf the amount of base needed to react with all the acid has been addedEnough base to react with all of the acid has been addedExcess base has been added.
This is because the anion of the weak acid becomes a. The OH ions from the strong acid combine with acidic ions in the buffer. If the pH of the solution is less than 7 is said to be an acidic solution.
Yes hydorchloric acid HCl is a strong acid. NaOH is an Arrhenius base because it increases the concentration of hydroxide ions when dissolved in the solution. HCl is the acid.
QUESTION 5 Which of the following statements describes a strong acid. NAWhich of the following describes the following titration curve. Which of the following criteria accurately describes the primary difference between a strong versus weak acid.
Strong acids and strong bases completely dissociate in. It is a strong acid. NaOH is the acid.
There is a sharp increase in pH at the beginning of the titration. See answers 2 Best Answer. View this answer View a sample solution Step 2 of 5.
HCI Hydrochloric acid Wiki User. Have a strong conjugate base.
Acid Base Reactions Boundless Chemistry

Acids And Bases I Introduction
What Makes Nh3 A Strong Acid Quora

Strong Acid Strong Base Reactions Video Khan Academy

What Are Strong Acids Or Bases Are That Ionize Or Dissociate In Aqueous Solution Socratic
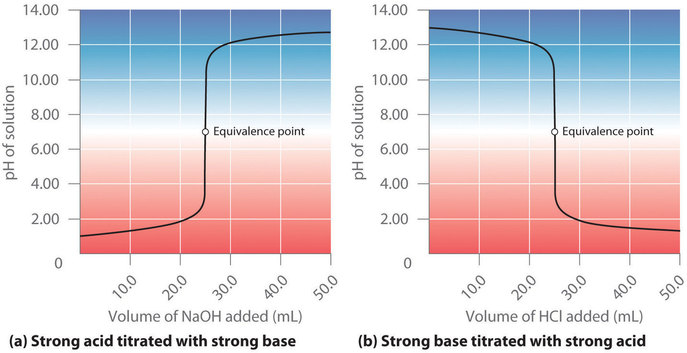
7 3 Acid Base Titrations Chemistry Libretexts
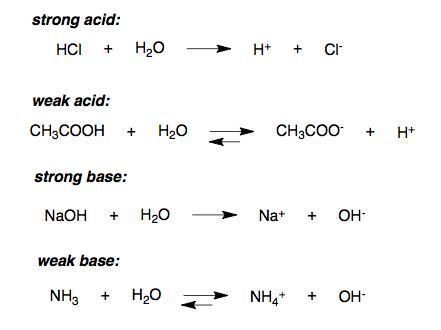
Strong Acids Introduction To Chemistry
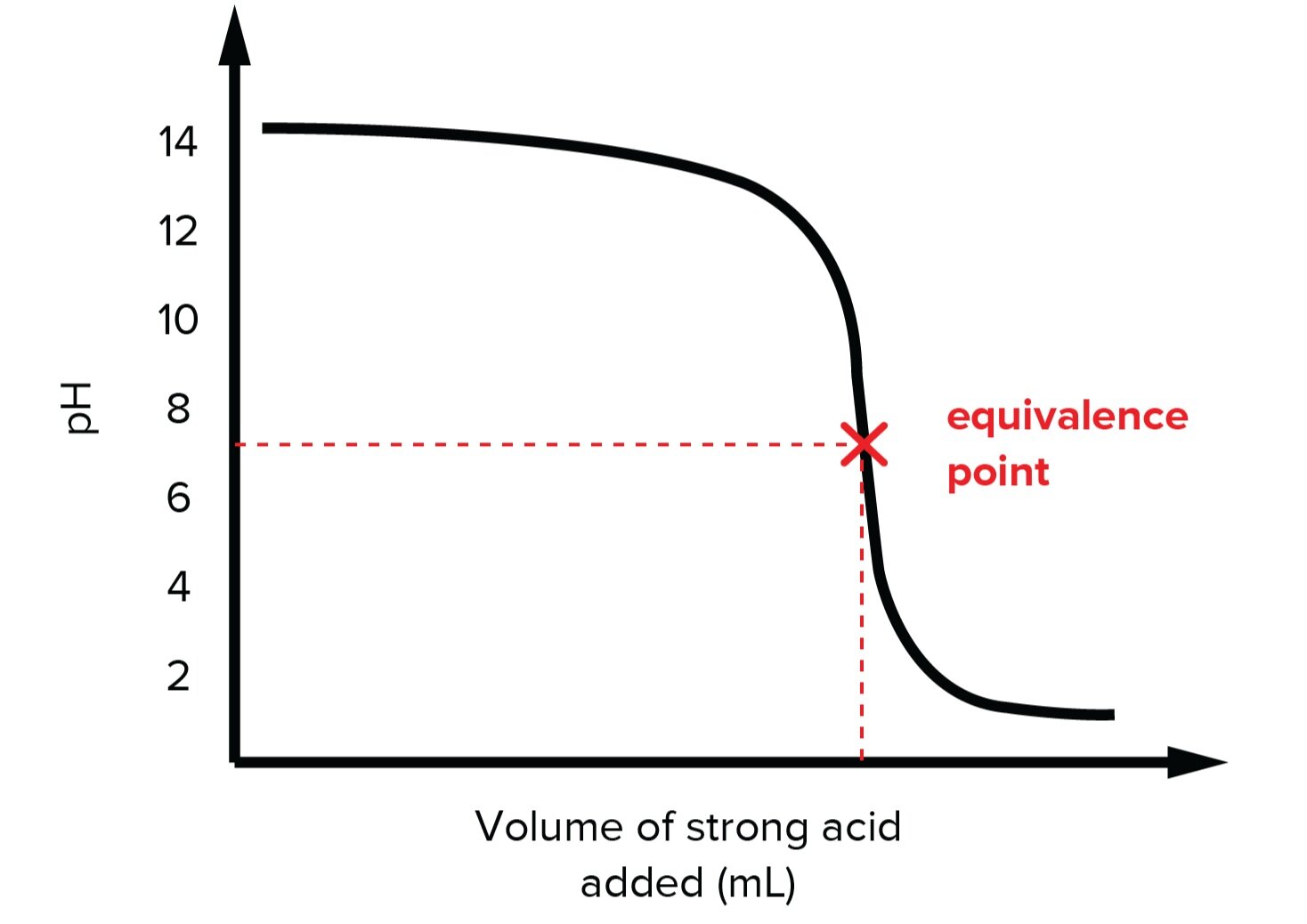
Acids And Bases For The Mcat Everything You Need To Know Shemmassian Academic Consulting
0 Response to "Which of the Following Describes a Strong Acid"
Post a Comment